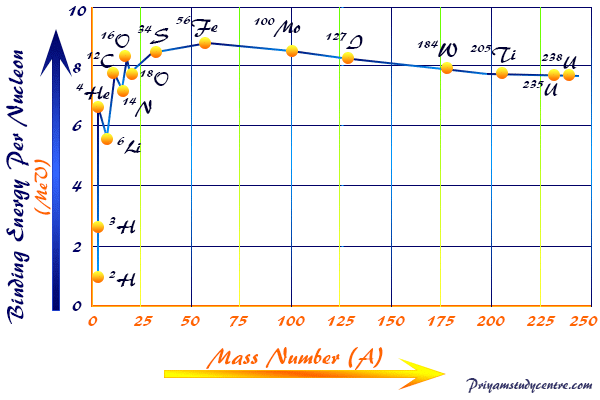
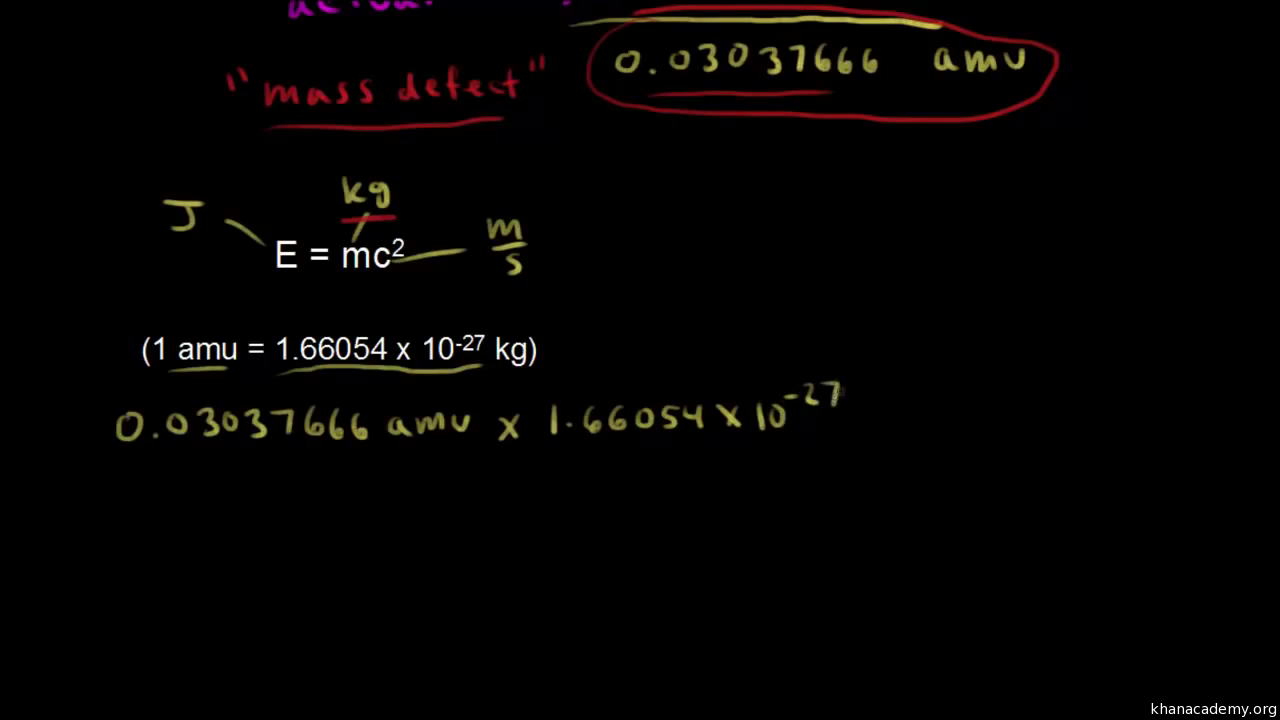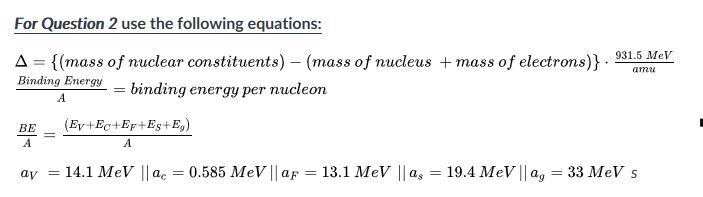SOLVED: Binding Energy perNucleod mass number (# of protons neutrons) 1) Calculate mass defect protons: 1.007276 amu neutrons: 1.008665 amu atomic number electrons: 0.0005486 amu (# of protons) 2) Convert amu kg

Mass Defect Formula & Examples | What is Nuclear Mass Defect? - Video & Lesson Transcript | Study.com
Calculate mass defect and binding energy per nucleon of 2010Ne, given. - Sarthaks eConnect | Largest Online Education Community
![Calculate the mass defect and the binding energy per nucleon of the ""(47)^(108)Ag nucleus. [atomic mass of Ag = 107.905949] Calculate the mass defect and the binding energy per nucleon of the ""(47)^(108)Ag nucleus. [atomic mass of Ag = 107.905949]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/378267553_web.png)
Calculate the mass defect and the binding energy per nucleon of the ""(47)^(108)Ag nucleus. [atomic mass of Ag = 107.905949]

Calculate the (i) mass defect, (ii) binding energy and (iii) the binding energy per nucleon for a 6C^12 nucleus. Nuclear mass of 6C^12 = 12.000000 a.m.u., mass of hydrogen nucleus = 1.007825

Calculate the mass defect of the helium nucleus 42He. The mass of neutral 42He is given by MHe=4.002603u. - Brainly.com