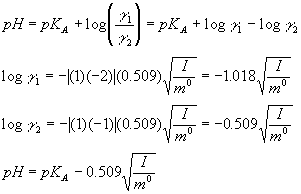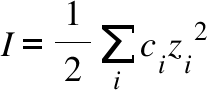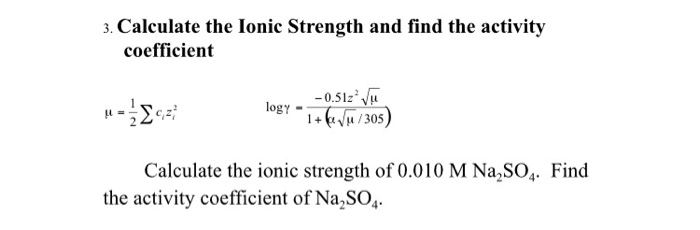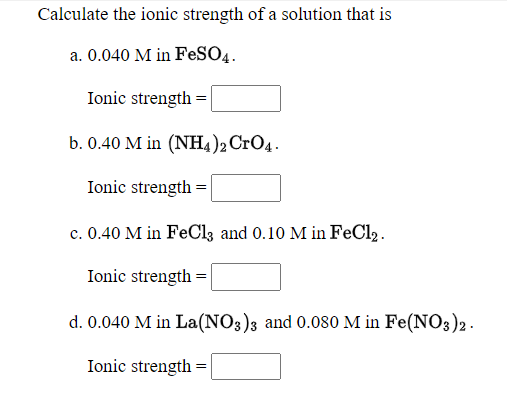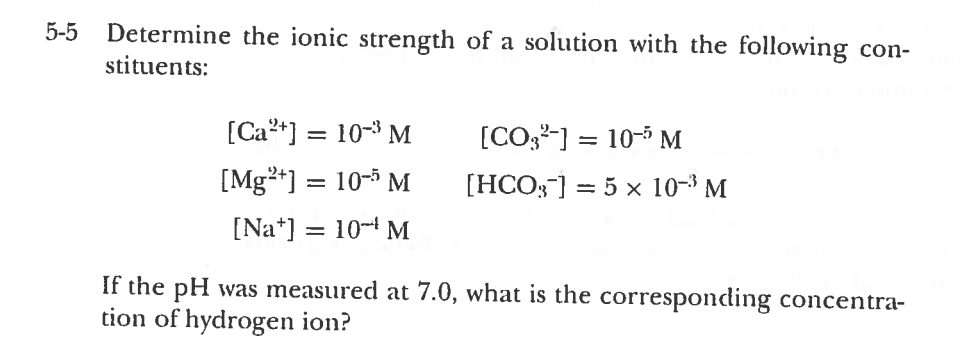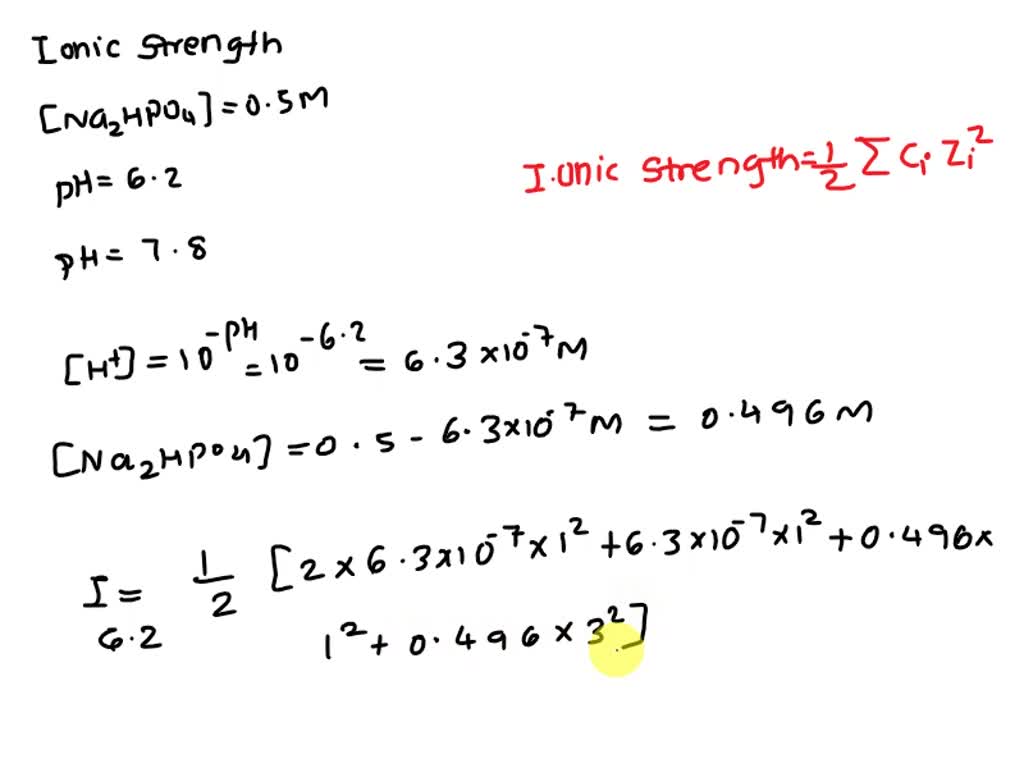Calculate the ionic strength of major ions of: a) coastal seawater of salinity 17.5 (i.e. one half of typical open ocean salinity) in mol kg-1 b) 0.7 mol/l NaCl c) another solution

25.4 g of iodine and 14.2 g of chlorine are made to react completely to yield a mixture of ICl and ICl3 . Calculate the number of moles of ICl and ICl3 formed.
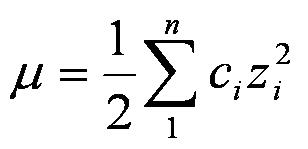
pH calculations and more in fundamentals of pharmaceutics. : What is ionic strength of solutions and how is it calculated?