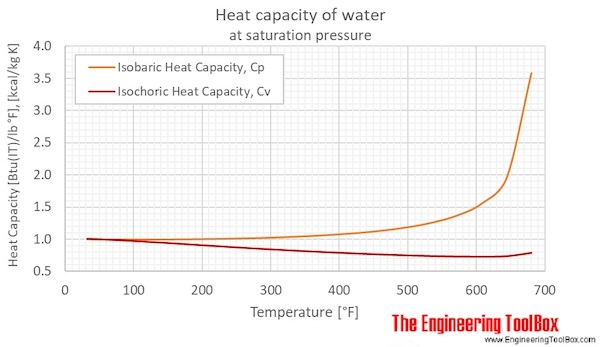
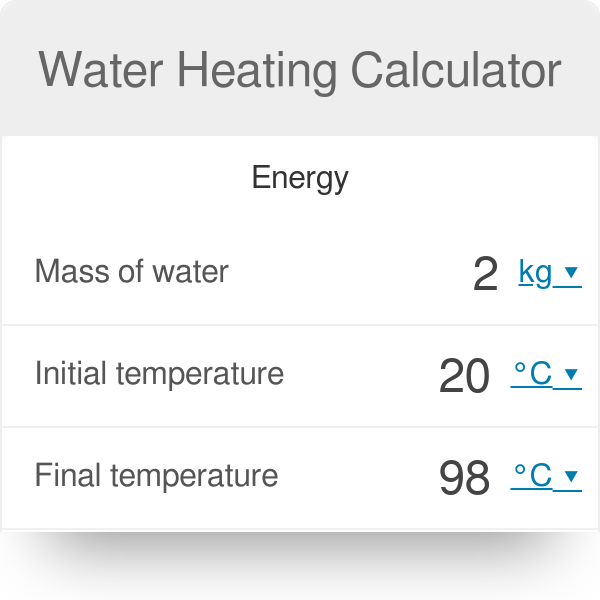
200 g of hot water at 80∘ C is added to 300 g of cold water at 10∘ C. Calculate the final temperature of the mixture of water. Consider the heat taken

ratio - Equation to calculate percentage of $\color{red}{\text{hot}}$ water needed to raise $\color{blue}{\text{cold}}$ water to $x^\circ$ using mixing valve - Mathematics Stack Exchange

20g of hot water at 80°C is poured into 60g of cold water, when the temperature of cold water rises by - Brainly.in

200 g of hot water at 80^@C is added to 300 g of cold water at 10^@C. Neglecting the heat taken by the container, calculate the final temperature of the mixture of

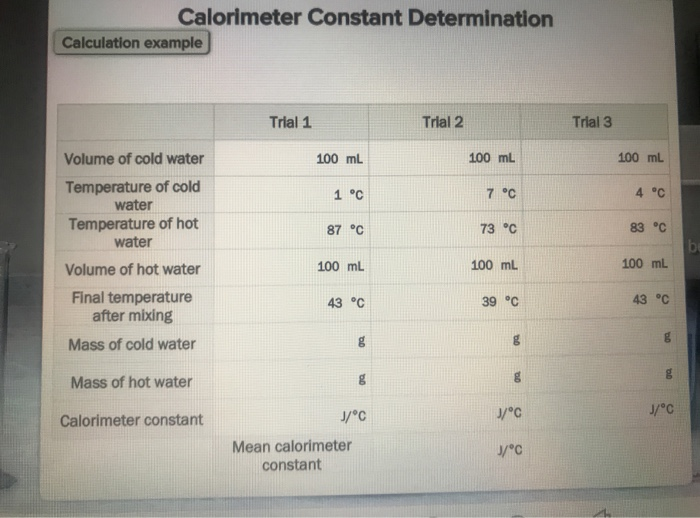
4.05 Physics - Virtual Lab Simulation Questions: 1. Include a data table that organizes the data collected from the three trials. Trial Volume of Cold | Course Hero
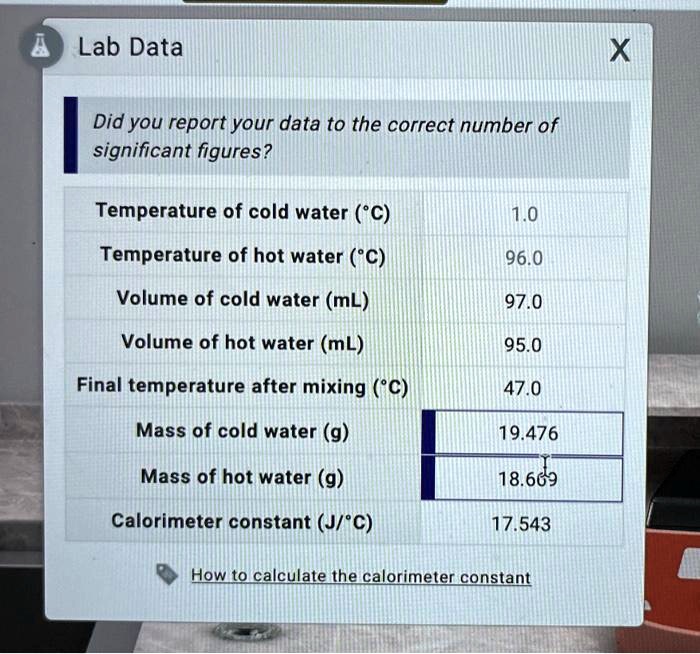
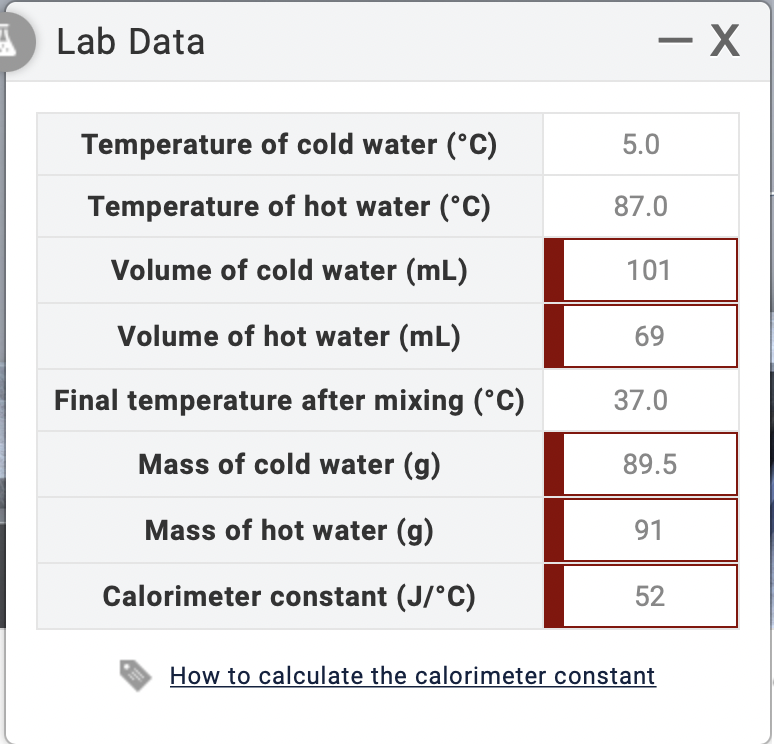
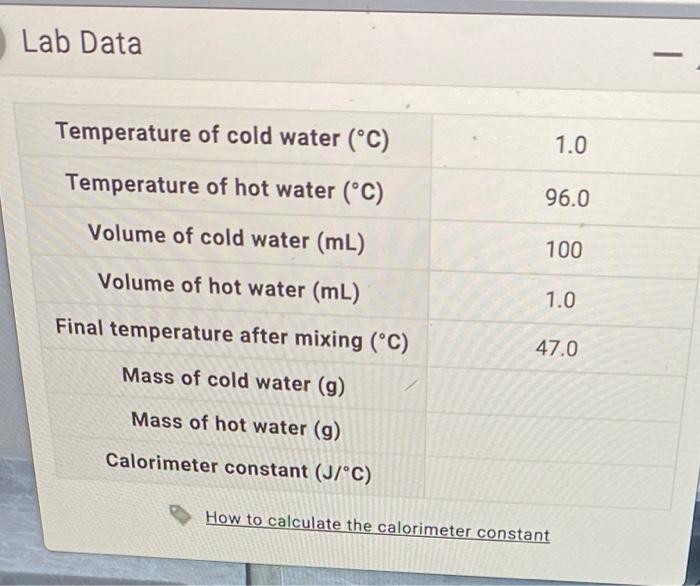
SOLVED: Lab Data Did you report your data to the correct number of significant figures? Temperature of cold water ("C) Temperature of hot water ('C) Volume of cold water (mL) Volume of
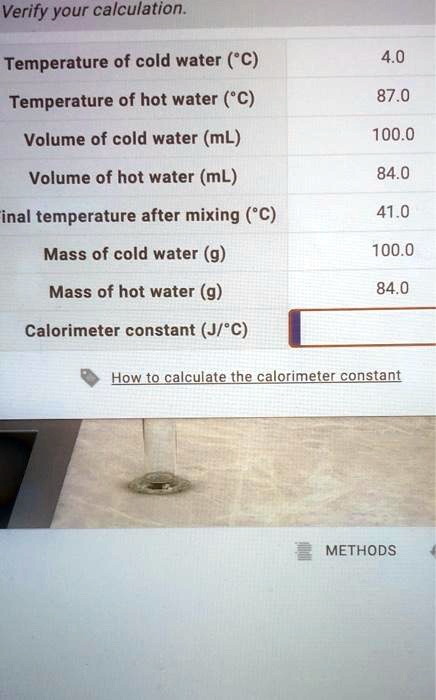
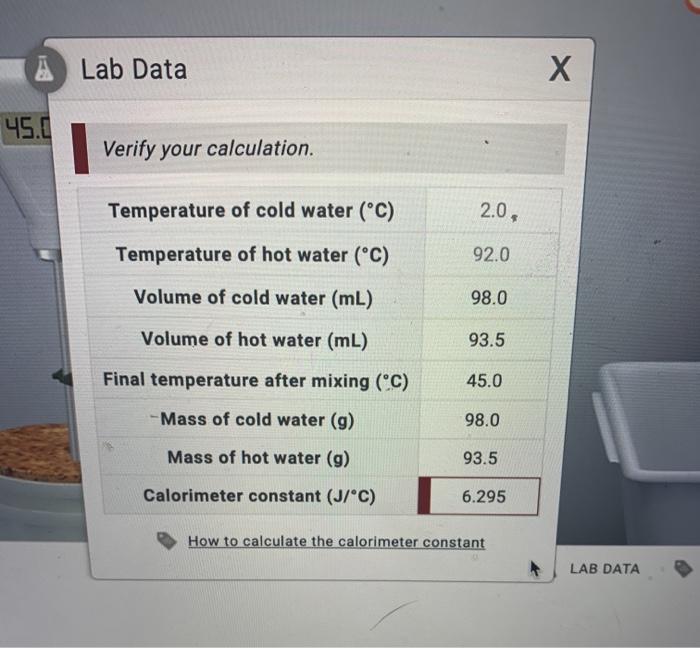
SOLVED: Verify your calculation: Temperature of cold water ("C) Temperature of hot water ("C) Volume of cold water (mL) 4.0 87.0 100.0 Volume of hot water (mL) 84.0 inal temperature after mixing ('

SOLVED: Lab Data Temperature of cold water (*C) Temperature of hot water ('C) Volume of cold water (mL) Volume of hot water (mL) Final temperature after mixing ('C) Mass of cold water (