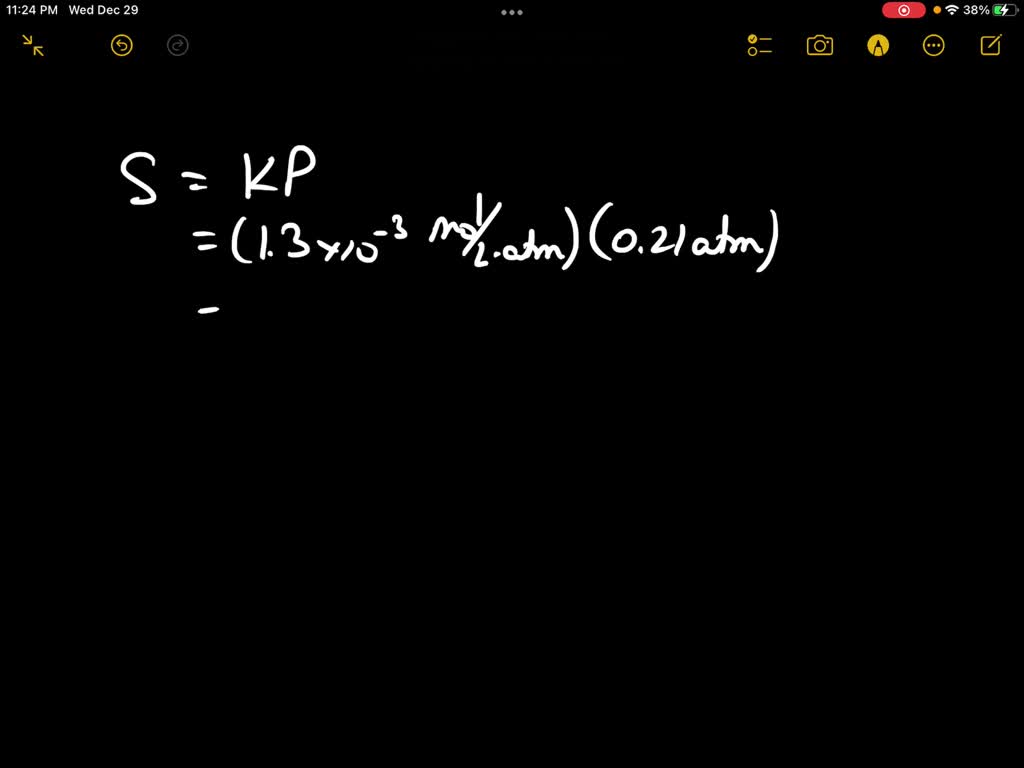
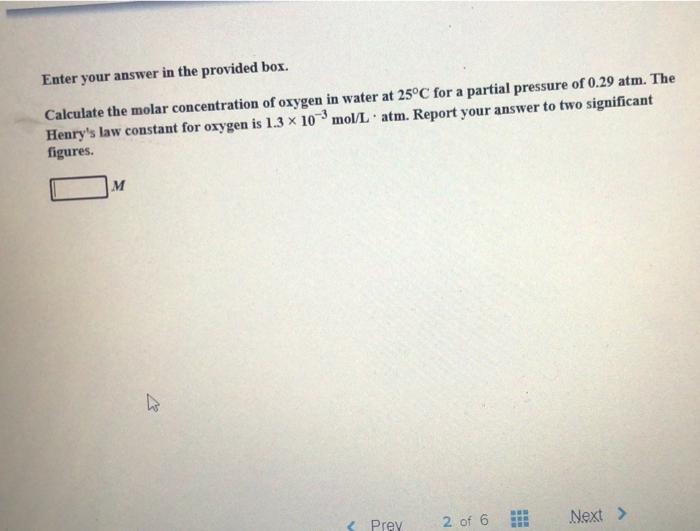
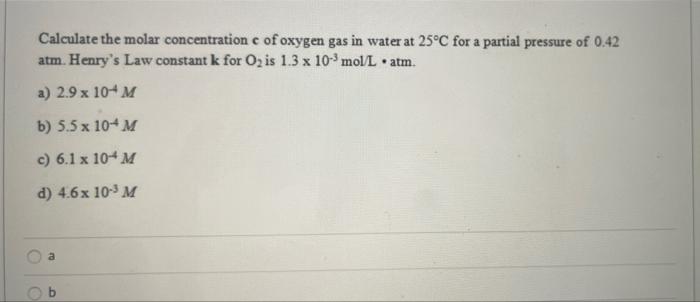
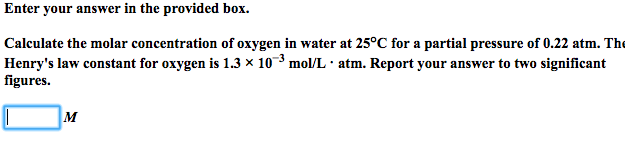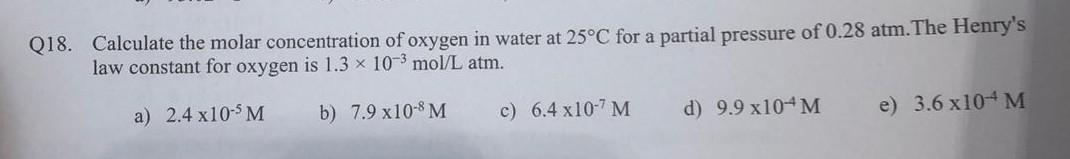SOLVED: Calculate the molar concentration of oxygen in water at 25°C for a partial pressure of 0.24 atm. The Henry's law constant for oxygen is 1.3 × 10−3 mol/L · atm. Report

The Henry's law constant for oxygen gas in water at `25^(@)C` is `1.3xx10^(-3)` M `atm^(-1)`. - YouTube
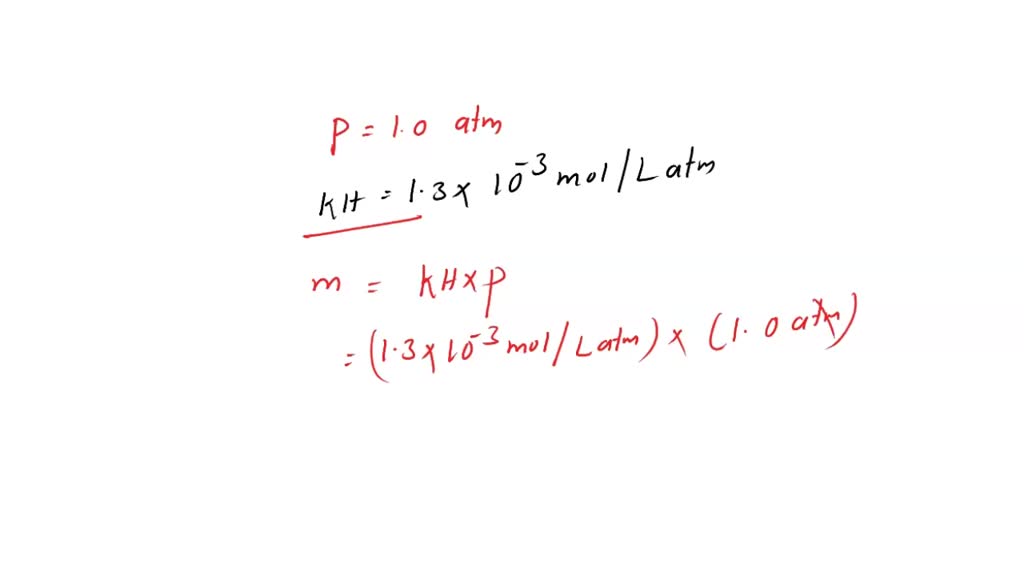
SOLVED: What is the molar concentration of oxygen in water at 25C for a partial pressure of 1.0 atm (KH = 1.3 x 10-3 mol/Latm)
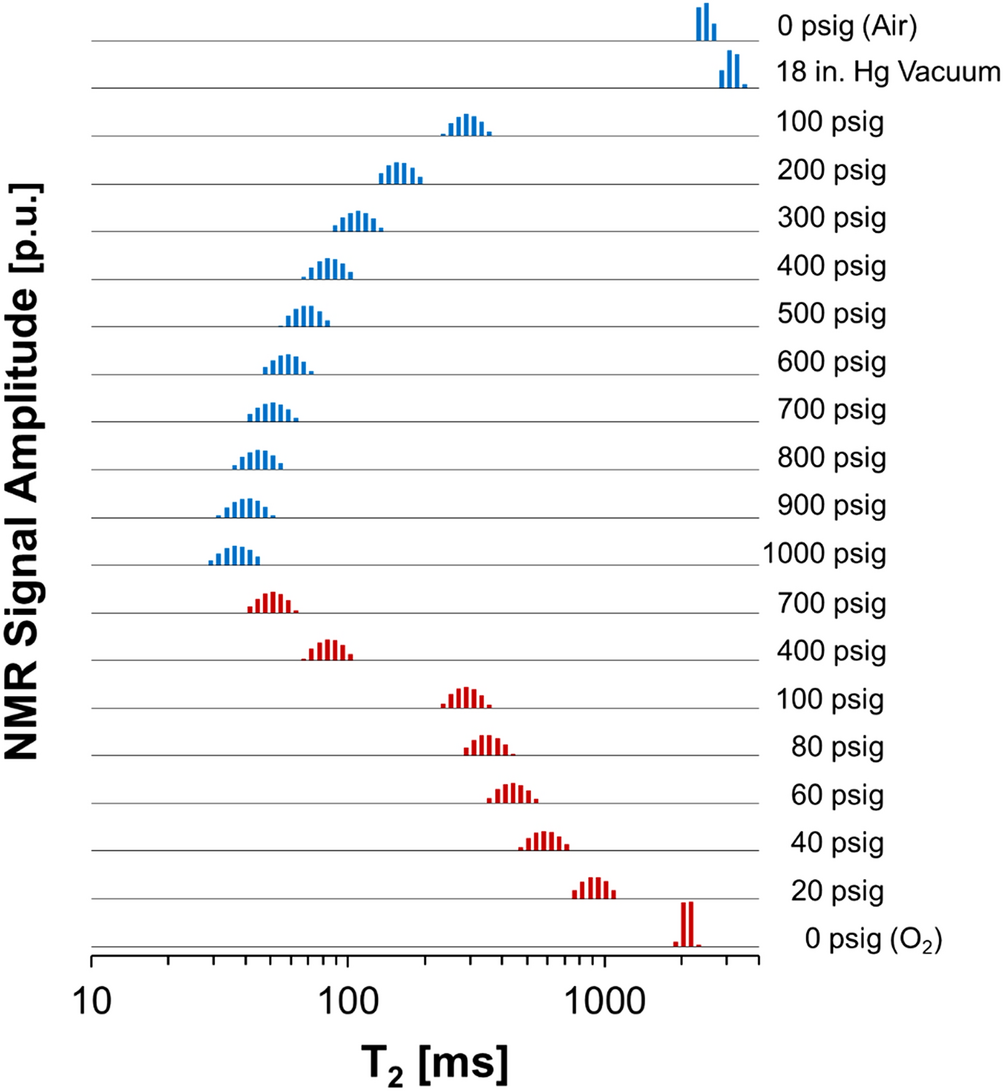
Quantification of dissolved O2 in bulk aqueous solutions and porous media using NMR relaxometry | Scientific Reports

Henry's law constant for oxygen dissolved in water is 4.34 × 10^4 atm at 25^C . If the partial pressure of oxygen in air is 0.4atm. Calculate the concentration (in moles per

SOLVED: Enter your answer in the provided box Calculate the molar concentration of oxygen in water at 259€ for partial pressure of 0.H4 atm . The Henry'haw constant for oxygen is 13

SOLVED: Enter your answer in the provided box: Calculate the molar concentration of oxygen in water at 258C for a partial pressure of 0.27 atm: The Henry's law constant for oxygen is